Acute Systemic Toxicity
Acute systemic toxicity tests are conducted to measure a chemical’s capacity to cause harm or death within two weeks of a single, short-term exposure.
Animal Tests
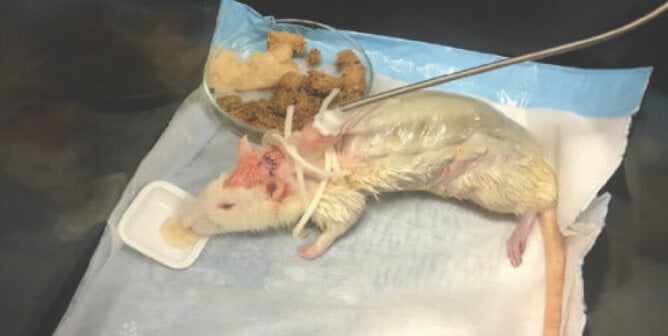
Mammalian acute systemic toxicity tests are commonly conducted on rats. The animals are exposed to a chemical, and based on the amount that kills them, an estimation of the amount that would kill half of them (the LD50) is made. The chemical is administered in high doses via the following routes.
- Oral: A chemical is pumped directly into an animal’s stomach via a force-feeding tube.
- Inhalation: Animals are forced to inhale the chemical while confined to a small tube.
- Dermal: A chemical is applied to an animal’s skin.
Acute systemic toxicity testing can have a number of effects on the animals, including severe abdominal pain, diarrhea, convulsions, seizures, paralysis, and bleeding from the nose, mouth, and genitals, before they ultimately die.
In addition to causing animals suffering, these tests are not reliable. For example, a study examining almost 2,500 chemicals showed that, when repeated, the oral animal test failed to produce the same result 40% of the time.
Non-Animal Tests
Mammalian acute systemic toxicity can be studied using computer models or human cells that assess the actions of toxic chemicals at the cellular level. For example, for acute dermal toxicity, three-dimensional reconstructed human skin models can be used first to determine whether a substance will penetrate the skin, which is necessary for a substance to cause systemic (dermal) toxicity.
For acute oral toxicity, there are computational models such as CATMoS (the Collaborative Acute Toxicity Modeling Suite) that can predict toxicity following oral consumption of a substance. Additionally, the cell-based neutral red uptake cytotoxicity test and the human cell–based AcutoX test can help predict if a substance is not toxic.
For acute inhalation toxicity, three-dimensional models constructed from human cells (such as those from Epithelix Sàrl) can be used to study the effects of chemicals on specific parts of the lung. In addition, “lung-chips”—which include relevant cell types and even mimic the breathing motion of the lung—can be used to model the human lung.
To assess the acute systemic toxicity of products comprising a number of different ingredients, the GHS additivity equation can be used to consider the individual toxicity values of each ingredient to predict the overall toxicity of the product.
PETA scientists have hosted workshops and webinars, published articles in peer-reviewed scientific journals, and organized funding and testing studies with the goal of developing strategies to replace animal use in acute systemic toxicity testing.