Background Information on Publicly Funded Animal Experiments in Virginia
Learn how agencies responsible for overseeing the use of animals in experiments fail to protect them, why testing on animals is ineffective, and how many animals suffer in Virginia in the name of science.
- Oversight Systems
- The 3Rs
- Congress Passed the FDA Modernization Act
- Why Noncompliant Experiments Produce Poor Data
- Number of Animals Used
- Funding
- Media Coverage
- Helpful Links
Oversight Systems
The federal Animal Welfare Act (AWA), which was passed in 1966, is the only federal law protecting animals kept in laboratories for use in experiments. It stipulates only meager, survival-level care standards, and dozens of species are excluded, among them those most commonly experimented on: coldblooded species (amphibians and reptiles), fish, invertebrates (including crustaceans and insects), birds, rats of the genus Rattus, and mice of the genus Mus, who are “bred for use in research.” There is no federal requirement to report the number of such animals used, nor is there one to report on efforts to adopt available non-animal testing methods.
Two federal agencies—the U.S. Department of Agriculture’s (USDA) Animal and Plant Health Inspection Service (APHIS) and the National Institutes of Health’s (NIH) Office of Laboratory Animal Welfare (OLAW)—are responsible for ensuring that facilities comply with federal animal welfare standards. These entities make up an imperfectly overlapping list, since in most cases, an individual organization is overseen by both agencies:
- A facility would be under APHIS oversight if it used AWA-covered species but didn’t receive funding from the National Institutes of Health (NIH).
- A facility would be under OLAW oversight if it received NIH funding for activities involving animals but none of the animals were AWA-covered species.

APHIS
APHIS is responsible for enforcing the AWA. However, the USDA’s Office of Inspector General has conducted several audits concluding that APHIS oversight of animal testing facilities is essentially meaningless. Even when animals have suffered or died due to clear negligence, the USDA systemically fails to penalize institutions sufficiently—or at all—for violations of the AWA.
As was the case with Envigo, a federally licensed entity can repeatedly and severely violate the AWA—even receiving USDA citations by the dozens—and chronically continue to fail to improve conditions for animals. Repeat offenders are allowed to operate while they’re in serious—even critical—and repeated violation of federal law.
All APHIS-covered facilities must register with the USDA, submit annual reports, and undergo USDA inspections, which may produce documentation of AWA violations. Annual reports, inspection reports, and some enforcement actions (which are rare and typically issued only for repeat or especially egregious violations) are publicly available, either on the APHIS website or through a Freedom of Information Act (FOIA) request to APHIS (which can take months, even years, to fulfill).
OLAW
OLAW oversees all public and private facilities that receive NIH funding for activities involving any vertebrate animals per the Health Research Extension Act of 1985 and the Public Health Service (PHS) Policy on Humane Care and Use of Laboratory Animals. A facility’s PHS Assurance (or registration) includes average daily inventory lists of species and descriptions of the Institutional Animal Care and Use Committee’s (IACUC) makeup. All facilities must have an IACUC (see below) as specified by the PHS Policy. Facilities are required to self-report violations of the PHS Policy to OLAW voluntarily. A facility’s PHS Assurance and OLAW correspondence regarding violations are publicly available via a FOIA request to OLAW.
The PHS Policy follows the Guide for the Care and Use of Laboratory Animals, which endorses, among other principles, “consideration of alternatives (in vitro systems, computer simulations, and/or mathematical models) to reduce or replace the use of animals.” It encourages researchers and their IACUC to consider the “availability or appropriateness of the use of less invasive procedures, other species, isolated organ preparation, cell or tissue culture, or computer simulation” and states that “the IACUC is obliged to weigh the objectives of the study against potential animal welfare concerns. By considering opportunities for refinement, the use of appropriate non-animal alternatives, and the use of fewer animals, both the institution and the principal investigator can begin to address their shared obligations for humane animal care and use.”
IACUCs
IACUCs are oversight bodies within individual institutions that are responsible for ensuring that animal use protocols adhere to the AWA and/or the PHS Policy.
A deviation from an IACUC-approved protocol is a violation of the AWA if it involves AWA-covered species and should be cited in an inspection report. It is a violation of the PHS Policy if it involves a vertebrate animal used in NIH-funded activities and should be reported to OLAW.
IACUC protocols can be obtained via FOIA requests from public institutions but not private ones.
AAALAC
The Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International is a private organization that offers an expensive “accreditation” program for facilities. Experimenters purport that its role is to “improve the welfare of animals produced for or used in research, teaching and testing, and to enhance the quality of these activities through accreditation of the animal care and use program.” A facility’s AAALAC accreditation primarily signifies that it pays an annual fee to the organization. Inspection reports are not made public, and facilities are only visited—not inspected—every three years.
AAALAC’s Council on Accreditation evaluates programs, approves the accreditation of facilities, and is made up of experts in the fields of “laboratory animal science or animal research.” The council members are experimenters themselves, which calls into question the integrity of the accreditation process. For example, one study showed that “AAALAC-accredited sites had significantly more AWA NCIs [noncompliant items] on average compared with non-accredited sites. AAALAC-accredited sites also had more NCIs related to improper veterinary care, personnel qualifications, and animal husbandry. These results demonstrate that AAALAC accreditation does not improve compliance with regulations governing the treatment of animals in laboratories.”
The 3Rs
The principle of the 3Rs (replacement, reduction, and refinement) was introduced in 1959 by W.M.S. Russell and R.L. Burch in The Principles of Humane Experimental Technique. Russell and Burch were members of the Universities Federation for Animal Welfare, a nonprofit organization with the mission “to diminish, by methods appropriate to its special character as a university organization, the sum total of pain and fear inflicted by man on animals.” The 3Rs principle advocates for alternatives to the use of animals in scientific studies (replacement), lowering the number of animals used (reduction), and changes that make the procedures performed on animals less distressing and/or painful (refinement).

The 3Rs principle has formally been adopted by practically every entity that uses animals for research and testing, forming the basis for legislation and regulations that govern the use of animals for this purpose. However, there is currently no mechanism in place to measure what, if any, action is taken by a facility to achieve the goals of the 3Rs principle.
Congress Passed the FDA Modernization Act
The bipartisan U.S. Food and Drug Administration (FDA) Modernization Act 2.0, cosponsored by senators Rand Paul, M.D. (R-KY) and Cory Booker (D-NY), was passed in 2022, allowing the FDA to approve drugs based on human-relevant, non-animal testing methods and removing the mandate on animal testing that had been in place since 1938. This paved the way for pharmaceutical companies to use modern, effective methods, such as computer modeling and organs-on-chips.
“The list of people and organizations celebrating [the FDA Modernization Act 2.0] modifying a longstanding FDA mandate on animal testing is long, broad, and bizarrely eclectic. It may be unprecedented for a proposal in Congress to make bedfellows of Rand Paul and Bernie Sanders, People for the Ethical Treatment of Animals (PETA) and the Cato Institute, and famed primatologist-turned-conservationist Jane Goodall and Texas congressman Marc Veasey, the former vice-chair of the pro-hunting Congressional Sportsmen’s Caucus …. Its fans include both Democrats and Republicans, high-profile individuals, and groups including the Crohn’s and Colitis Foundation, the National LGBT Cancer Network, the National Hispanic Medical Association, and at least 117 organizations devoted to animal rights.”
—Emily Sohn, “What If We Didn’t Have to Test New Drugs on Animals?”
This historic legislation reflects scientists’ current understanding regarding the inadequacies of animal “research models” and the fact that they can now be replaced by superior methods. Animals aren’t the best models for determining whether drugs are safe and effective for humans—nor are they the best models for learning about human diseases and developing drugs for them in the first place.
In the words of Sen. Paul, “The FDA Modernization Act 2.0 will accelerate innovation and get safer, more effective drugs to market more quickly by cutting red tape that is not supported by current science …. The inclusion of this bipartisan effort is a step toward ending the needless suffering and death of animal test subjects—which I’m glad both Republicans and Democrats can agree needs to end.”
The 2023 federal budget also includes $5 million for a program aimed at reducing animal testing by helping to develop and encourage industry to adopt new testing methods. To track the Commonwealth’s progress and to incentivize innovation and advances in research and development, we must establish a data baseline regarding the extent to which animals are used in experiments.
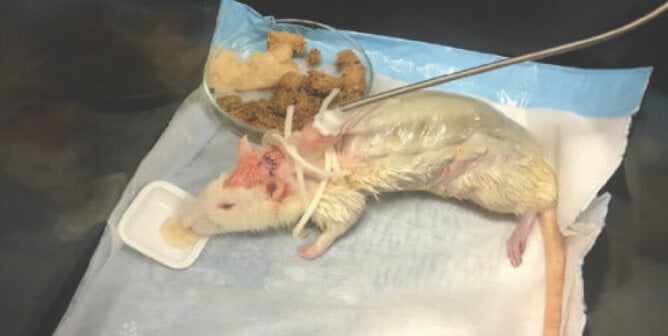
Why Noncompliant Experiments Produce Poor Data
Even when animal experiments comply with the virtually nonexistent standards and regulations that are in place, the majority of the studies don’t contribute to improving human health, and the value of the role that animal experimentation plays in most medical advances is questionable. It is well established that when animals in laboratories endure pain, inadequate housing, improper food and water provisions, improper temperature, or irregular lighting or become injured and/or suffer from stress during capture and restraint, their physiology and behavior are drastically altered. As a result, any data gathered from them becomes even further compromised and unreliable. Deviations from animal welfare standards not only are ethically problematic but also produce results that are scientifically useless and/or misleading. They result in wasted funds and time, and they harm animals with absolutely no justification whatsoever.
Numerous scientific studies and reviews reveal that experiments on animals—even when they’re conducted in accordance with the minimal animal welfare standards that exist—fail to lead to effective treatments and cures for human diseases, including the top killers in the U.S. Critically, intrinsic biological and genetic differences among species contribute significantly to inescapable problems in extrapolating results from nonhuman animals to humans, even in the best-controlled and best-executed study designs. NIH reports that novel drugs fail “in about 95 percent of human studies,” even though they appeared safe and effective in preclinical experiments using animals. Reliance on animal models is diverting funds away from more promising areas of research and delaying the development of effective drugs and treatments as well as limiting the ability to protect human and environmental health.
To learn more about why animal testing is bad science, click here.
Number of Animals Used
According to the USDA’s 2024 annual reports, 7,051 animals covered by the AWA were used in Virginia testing facilities in that year alone. However, this number represents only approximately 5% of the animals actually used, since most species are not protected by the act.
There are currently no reporting requirements for the majority of species used in experiments at publicly funded Virginia institutions. The number of animals kept and used at facilities should be a matter of public record in order to ensure some measure of transparency and accountability and to safeguard public trust.
The USDA’s 2024 annual reports show that the following animals were held or used in experiments at publicly funded Virginia institutions.
| Type of Animal | Animal Counts |
| Dogs | 73 |
| Cats | 41 |
| Guinea pigs | 133 |
| Hamsters | 105 |
| Rabbits | 320 |
| Nonhuman primates | 29 |
| Pigs | 359 |
| Other* | 5,955 |
| Total AWA-covered animals | 7,051 |
*Other species include the following:
- Domestic horses (38)
- Common tree shrews (37)
- Domestic ferrets (10)
- Birds
- White-crowned sparrows (392)
- House finches (703)
- Ruminants
- Cattle/cows/oxen/Watusis (113)
- Domestic goats (3)
- Rodents
- Marsh rice rats (8)
- Western deer mice (298)
- Mongolian gerbils (57)
- Seba’s short-tailed bats (20)
- Jamaican fruit bats (105)
- Groundhogs or woodchucks (3)
- Prairie voles (4,168)

Animal testing facilities are required to categorize the animals they use for research in their annual reports as follows:
- Column B—Animals held by a facility but not used in any research that year
- Column C—Animals used in research; no pain involved; no pain drugs administered
- Column D—Animals used in research; pain involved; pain drugs administered
- Column E—Animals used in research; pain involved; no pain drugs administered
- Facilities must provide explanations for these “animals upon which teaching, experiments, research, surgery, or tests were conducted involving accompanying pain or distress to the animals and for which the use of appropriate anesthetic, analgesic, or tranquilizing drugs would have adversely affected the procedures, results, or interpretation of the teaching, research, experiments, surgery, or tests.”
- According to USDA annual reports, in 2021, Virginia animal testing facilities reported using 682 animals in this category. However, this number represents only approximately 5% of the animals actually used, since most species aren’t protected by the AWA.
Funding
In 2024, the Commonwealth of Virginia and its localities gave public universities in the state nearly $180 million for animal and non-animal research.
| Institution | State/Local Sources | Federal Sources | All Sources |
| Christopher Newport University | $5,000 | $2,224,000 | $3,764,000 |
| Eastern Virginia Medical School | $676,000 | $21,658,000 | $27,891,000 |
| George Mason University | $3,573,000 | $147,327,000 | $281,697,000 |
| James Madison University | $1,814,000 | $3,482,000 | $21,394,000 |
| Norfolk State University | $313,000 | $4,918,000 | $7,309,000 |
| Old Dominion University | $13,307,000 | $32,045,000 | $76,202,000 |
| University of Mary Washington | $3,000 | $610,000 | $1,333,000 |
| University of Virginia’s College at Wise | $87,000 | $1,207,000 | $1,464,000 |
| University of Virginia | $24,266,000 | $394,731,000 | $813,772,000 |
| Virginia Commonwealth University | $47,241,000 | $208,743,000 | $524,295,000 |
| Virginia Tech | $80,644,000 | $308,282,000 | $656,916,000 |
| Virginia State University | $4,144,000 | $6,684,000 | $13,271,000 |
| William & Mary | $3,683,000 | $41,488,000 | $94,609,000 |
| Total | $179,756,000 | $1,173,399,000 | $2,523,917,000 |
The above figures include both animal and non-animal research. There is currently no available information indicating how much money is spent on animal experimentation in Virginia.
Media Coverage
- May 28, 2015: USA Today article “Universities, Feds Fight to Keep Lab Failings Secret”
- April 21, 2020: com article “Lawmakers Reject Northam Amendment That Could Have Increased Oversight of Controversial Dog Breeding Facility”
- July 21, 2022: Northeastern Global News article “4,000 Beagles Were Rescued From a Virginia Research Facility. How Common Is Lab Testing on Dogs in the U.S.?“
- October 2022: Life article “What if We Didn’t Have to Test New Drugs on Animals?”
- February 21, 2023: The Virginian-Pilot article “Virginia Bill Would Require Animal Testing Facilities to Submit Info to the State”
- March 6, 2023: The Virginian-Pilot article “Animal Welfare Advocates Disappointed With ‘Watered Down’ Testing Transparency Bill”
- March 28, 2023: The Virginian-Pilot article “Time’s Up: Gov. Youngkin Signed 738 Bills Into Law”
- January 28, 2025: “Virginia Senate passes bill requiring animal testing facilities to send unneeded primate to sanctuaries,” Courthouse News
- February 11, 2025: “Column: Primates deserve a chance at life after laboratory experiments,” The Virginian-Pilot
- February 19, 2025: “Senate, House approve bill urging better care for animals used in experiments,” Richmond Capital News
For a list of media coverage regarding the historic shutdown of Envigo’s dog-breeding factory and the related 2022 legislative activity, click here.